Comfortland CK-335 Ankle Support Support
Brand Comfortland Medical
Shop more from CFM
* Click "Add to Cart" to calculate shipping and delivery time.
Product Description
Manufacturer Part Number: CK-335
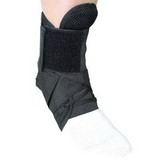
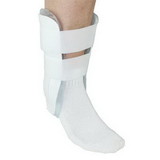
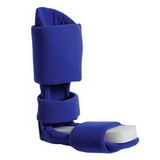
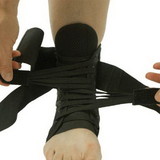
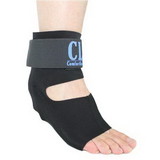
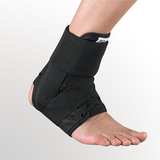
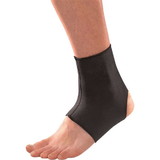
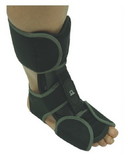
Designed to aid in drop foot that is secondary to CVA or peroneal nerve palsy as well as other neurological pathologies, the ankle support support is constructed from an injection-molded polypropylene, making it lightweight and allowing it to fit in most shoes. The CK-335 features a variable thickness design for added strength, with thinner areas at the front of the foot plate to allow for easy trimming, giving users a custom fit.
Application Instructions
PDAC Approval Letter
Brochure
Product Overview
Sugg HCPC: L1930
Indications
Peroneal paralysis
Acquired deformity of the foot and ankle
recovery Achilles care
Highlights
Injection molded polypropylene
Varying thickness for added strength
Trimmable footplate
Designed to aid in drop foot that is secondary to CVA or peroneal nerve palsy as well as other neurological pathologies, the ankle support support is constructed from an injection-molded polypropylene, making it lightweight and allowing it to fit in most shoes. The CK-335 features a variable thickness design for added strength, with thinner areas at the front of the foot plate to allow for easy trimming, giving users a custom fit.
Application Instructions
PDAC Approval Letter
Brochure
Product Overview
Sugg HCPC: L1930
Indications
Peroneal paralysis
Acquired deformity of the foot and ankle
recovery Achilles care
Highlights
Injection molded polypropylene
Varying thickness for added strength
Trimmable footplate
| Part # | Size |
| CK-335-3-L | Medium Left (10.5" support height) |
| CK-335-3-R | Medium Right (10.5" support height) |
| CK-335-4-L | Large Left (11.5" support height) |
| CK-335-4-R | Large Right (11.5" support height) |
 Attention CA Residents: Prop 65 Warning
Attention CA Residents: Prop 65 Warning
 WARNING:
This product can expose you to chemicals which are known to the State of California to cause cancer, birth defects, or other reproductive harm. For more information, go to www.p65warnings.ca.gov.
WARNING:
This product can expose you to chemicals which are known to the State of California to cause cancer, birth defects, or other reproductive harm. For more information, go to www.p65warnings.ca.gov.
Need more information?
Our staff are available to answer any questions you may have about this item
Our staff are available to answer any questions you may have about this item
Look Similar Items By Category